This product is manufactured by
MEKICS
Manufacturer information

MEKICS
Medical equipment manufacturer, Patient monitoring unit, Aritificial respirator, Ventilator, Pulse oximeter and more
Inquiry
How to order
Problem with product info?
Update request
Manufacturer
MEKICS
Product Type
Machine
Brand
-
SKU
31486
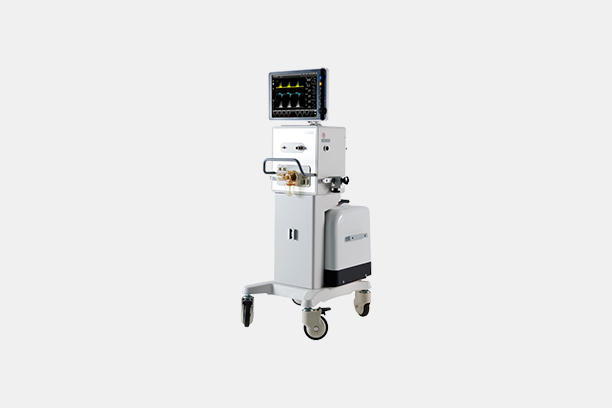
Product Name
CPAP
Model Name
M3
Size
-
Weight
-
Product Details